Moscow region, Odintsovo
st. Soyuznaya, 7
info@techmedmsk.ru
What is cleanliness? Determination of helium purity
What is purity? Determination of helium purity
When we talk about special gases, the concept of purity is mentioned quite often. But what is meant in this case by purity? How can you define it?
The purity of gases characterizes the presence of a certain amount of impurities in a substance. At the same time, specific sizes of limits for impurities are established, which should not exceed this value. In the case of using gases without specific impurities, they should be considered as typical. When filling cylinders or containers with high-purity gas, the tank is accompanied by the corresponding certificates of analysis, which confirm that the limit values of the impurity fractions are not exceeded. This document indicates the purity level of the gas in the tank.
This procedure does not apply to “Gases in small cylinders”.
The gas quality indicators located in the cylinder often indicate a higher level of purity than documented. However, this figure should not be taken as a guarantee of a higher purity of the substance.
Perhaps the simplest and most accurate definition for characterizing gas purity is: “Purity is the absence of impurities in a substance.”
Impurities can be understood as a material or substance in the finished product that differs from the chemical composition of the material or compound being sold. However, you need to know that in any case, the production process cannot provide absolute, 100% gas purity.
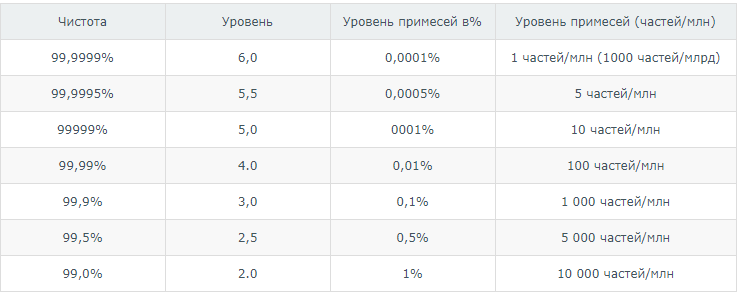
In classification, the cleanliness level is recorded in two ways:
- As a code, for example 5.5, where the digit before the decimal point indicates the number of nines, and the digit after the period indicates the last decimal place in the purity level value, for example: 5.5 = 99, 9995% or 6.0 = 99.9999%. < / li>
- Percentage like & gt; 99.9995
Examples:
– ethylene grade 2.8 means gas of 99.8% purity;
– argon grade 6.0 means 99.9999% purity;
Despite the purity stated in the product description, some gases with an additional prefix (for example – Nitrogen CO-free) have a particularly low residual content of certain impurities.




